Which One of the Following Is a Weak Acid
This is inherently unsatisfying because acid strengths are a continuum. Which of the following is a weak acid.

Solved Which One Of The Following Is A Weak Acid Hno 3 Hi Chegg Com
Which one of the following is a strong acid.
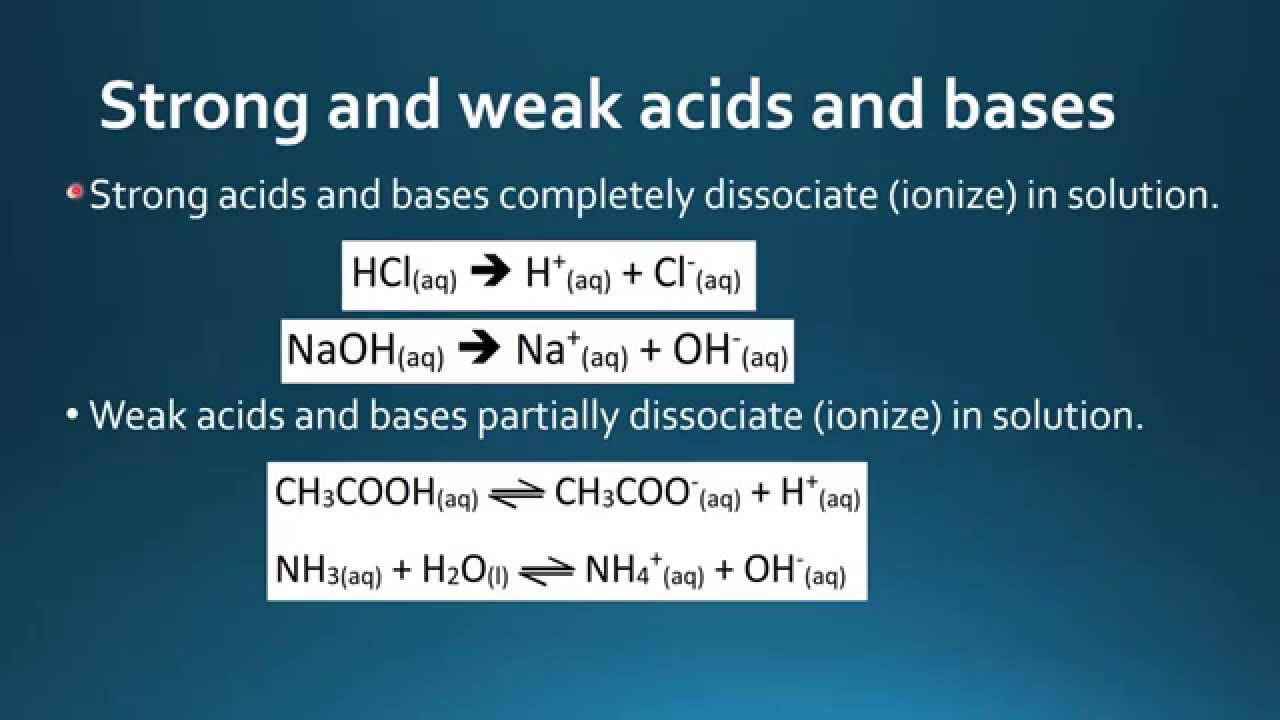
. CH 3 COOH is a weak acid because it ionizes only partially in aqueous solution to form H and CH 3 COO ions. 1 The hydrogen oxalate anion HC2O4- is a stronger base than the dihydrogen phosphate anion H2PO4-. Which one of the following is a weak acid.
A weak acid and its conjugate base in solution Question 9 A buffer solution contains ethanoic acid and its conjugate base. Sodium hydroxide O B. Most organic acids are weak acids.
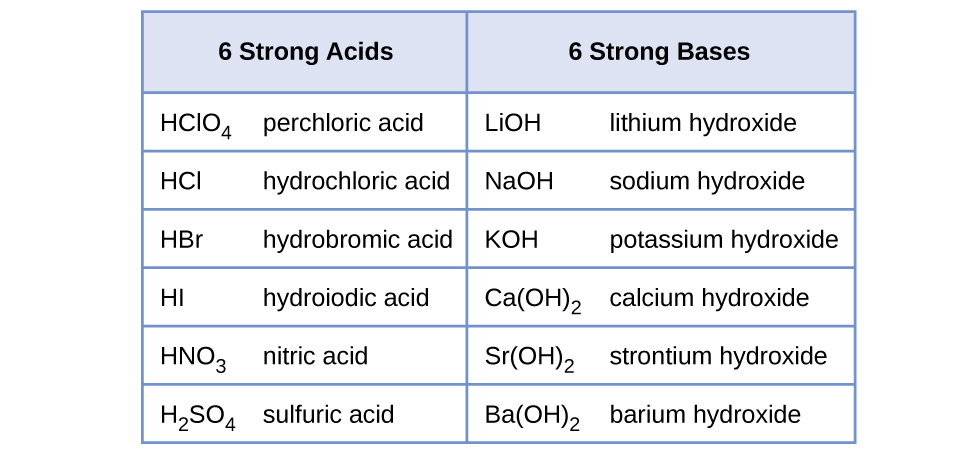
The strong acids are hydrochloric acid nitric acid sulfuric acid hydrobromic acid hydroiodic acid perchloric acid and chloric acid. CH 3 COOH CH 3 COO H. Which one is strong or weak or not.
The only weak acid formed by hydrogen and a halogen is hydrofluoric acid HF. Therefore the nature of CH 3 COO is basic and we call CH 3 COO the conjugate. Add your answer and earn points.
Which one of the following is a weak acid. A ethanoic acid b nitric acid csulphuric acid dhydrochloric acid. The chemical equation for the given question may be written as.
The substance CH3CH22NH is considered to be A. Weak acids are effective antimicrobials in their protonated form. Which one of the following is a weak acid.
Your own stomach utilizes hydrochloric acid to. In a weak acid this is not true and you will generally have most of the starting acid molarity still remaining at equilibrium. The only weak acid formed by the reaction between hydrogen and a halogen is hydrofluoric acid HF.
4-chlorobenzoic acid is a weak acid. Rejialeena84 rejialeena84 03032020 Chemistry Secondary School answered Which of the following is a weak acid. Click here to get an answer to your question Which one of the following is an example of a weak acid.
Neither acidic nor basic. Which of the following is a weak acid. Complete Self Study Package designed by.
CHEM 1311 EXAM 2docx - Which one of the following is a. A H2SO4 b HF c HCl d HBr e HI 1 See answer Advertisement Advertisement gldenps7911 is waiting for your help. Which of the following is a weak acid.
Which one of the following is a strong acid. H3PO4aq 3OH-aq PO43-aq 3H2Ol a If all the water evaporated away the salt remaining could possibly be Na3PO4. Common weak acids include acetic acid boric acid hydrofluoric acid oxalic acid citric acid and carbonic acid.
This preview shows page 1 - 4 out of 11 pages. Which among the following is a weak acid. Citric acid is a weak acid because it has a very small amount of H ions in its solution.
3 Oxalic acid is a weaker acid than phosphoric acid. This problem has been solved. B The acid H3PO4 is a weak electrolyte.
For such reason they are mostly used in the preservation of acid foods. Vinegar contains acetic acid. D is the sum of the potential and kinetic energies of the components.
The pH of the solution was 493 at 250 C. Lucyw17 lucyw17 The answer is b HF Advertisement Advertisement New questions in Chemistry. While technically a weak acid hydrofluoric acid is extremely powerful and highly corrosive.
A Its relative molecular mass b The pK b of its conjugate base. A ethanoic acid b nitric acid csulphuric acid dh Get the answers you need now. See the answer See the answer See the answer done loading.
4 Phosphoric acid is a. A HNO 3 B HCl C HI D HF E HClO 4. Home Community Solution A is prepared by dissolving 10 ml of hydrochloric acid in 100 ml of water and solution B is prepared by dissolving 1 ml of hydrochloric acid in 100 ml of water.
C The base involved must be a strong soluble base. HBr HCl HI HNO3 HClO4 H2SO4. Common strong acids include hydrochloric acid sulfuric acid phosphoric acid and nitric acid.
Acetic acid C H 3 C O O H Formic acid H C O O H Carbonic acid H 2 C O 3 are considered as weak acids. Design classes One-on-One in your own way with Top IITiansMedical Professionals Click Here Know More. Which one is more concentrated.
Strong acids have high K a or. E none of the above. What is weak acid used for.
Group of answer choices. It is found in citrus fruits such as lemons and oranges. HZ is a weak acid.
If we take the definition of strong acid as pKa 0 then a weak acid is one with a pKa 0. Which one of the following is a common acid vinegar. The strong acids are hydrochloric acid nitric acid sulphuric acid hydrobromic acid hydroiodic acid perchloric acid and chloric acid.
9 The balanced reaction between aqueous nitric acid and aqueous strontium hydroxide is. D This is classified as a neutralization reaction. Which among the following is a weak acid.
1 CH 3 COOH 2 H 2 SO 4 3 HCl 4 HNO 3. For example HF dissociates into the H and F ions in water. Which one of the following is equal to the pK a of a weak acid.
An aqueous solution of HZ is prepared by dissolving 0020 mol of HZ in sufficient water to yield 10 L of solution. 8 Which one of the following is a weak acid. Which of the following acid is a weak acid.
Which of the following is a weak acid. Weak acids do not completely dissociate into their ions in water. 2 Water is a stronger acid than either oxalic or phosphoric acids.
A HNO3 B HCl C HI D HF E HClO4 ------ Answer.

Acids And Bases I Introduction

Solved Question 3 Which One Of The Following Is A Weak Acid Chegg Com

Pin On Chemistry Analytical Chem

War Of The Acids In 2021 Next Generation Science Standards Teacher Manual War
14 3 Relative Strengths Of Acids And Bases Chemistry

8 3 3 Distinguish Between Strong And Weak Acids And Bases Youtube

8 3 3 Distinguish Between Strong And Weak Acids And Bases Youtube

Strong And Weak Acids Bases Video Khan Academy

Strong And Weak Acids And Bases

List Of Strong Weak Acids Bases Chemistry Basics Chemistry Molecules

Solved Which Of The Following Is A Weak Acid A Nitric Chegg Com

Solved Which One Of The Following Is A Weak Acid A Hf B Chegg Com






Comments
Post a Comment